Looking at Table 5.2. 1, you see that the pK a of carboxylic acids are in the 4-5 range, the pK a of sulfuric acid is -10, and the pK a of water is 14. Alkenes and alkanes, which are not acidic at all, have pK a values above 30. The lower the pKa value, the stronger the acid. Table 5.2. 1: Representative acid constants.
Organic Chemistry chapter 3 Flashcards | Quizlet
Chemistry 10th Edition ISBN: 9781305957404 Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste Publisher: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste Chapter1: Chemical Foundations Section: Chapter Questions Problem 1RQ: Define and explain the differences between the following terms. a. law and theory b. theory and

Source Image: quizlet.com
Download Image
EXPERT VERIFIED Step 1/9 Step 1: Identify the acids in the given list. The acids in the given list are OH, NH, OH, CI, and 3. Step 2: Determine the pKa values of each acid. The pKa values of the acids are not provided in the given list. Therefore, we cannot directly rank them based on pKa values.
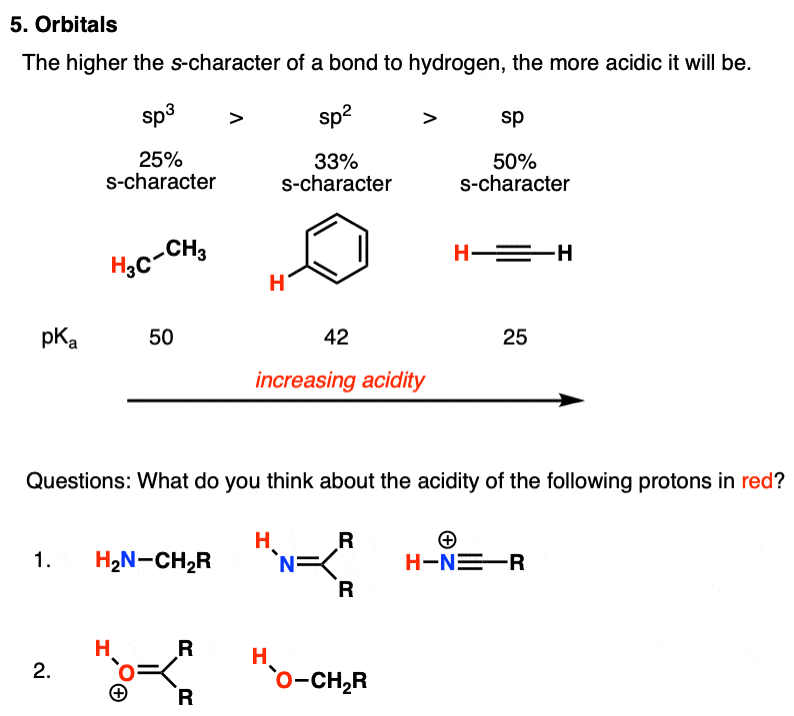
Source Image: masterorganicchemistry.com
Download Image
Question on pKa values for amino acids : r/Mcat Sep 11, 2023HO Submitted by Stacy P. Sep. 11, 2023 07:21 a.m. Instant Answer Step 1/3 Step 1: Determine the acidity of each acid based on its structure and functional groups. HO- is a hydroxide ion, which is a strong base and not an acid. Therefore, it does not have a pKa value. Step 2/3

Source Image: chem.ucalgary.ca
Download Image
Rank The Following Acids From Lowest Pka To Highest Pka
Sep 11, 2023HO Submitted by Stacy P. Sep. 11, 2023 07:21 a.m. Instant Answer Step 1/3 Step 1: Determine the acidity of each acid based on its structure and functional groups. HO- is a hydroxide ion, which is a strong base and not an acid. Therefore, it does not have a pKa value. Step 2/3 Predict the Outcome of Organic Acid-Base Reaction — Use pK a as Criterion With the knowledge of acidity and pK a, we are now ready to see how to apply this information to the understanding of organic reactions from an acid-base perspective.. The following reaction is an example in Section 3.2.If you take a closer look at the reactants and products, you will find that the “product” side
Ch21: Acidity of alpha hydrogens
3. Nitro groups are very powerful electron-withdrawing groups. The phenol derivative picric acid (2,4,6 -trinitrophenol) has a pK a of 0.25, lower than that of trifluoroacetic acid. Use a resonance argument to explain why picric acid has such a low pKa. [reveal-answer q=”475315″]Show Solution [/reveal-answer] Chemistry of Enolates and Enols – Acidity of Alpha-Hydrogens | Notes | PharmaXChange.info
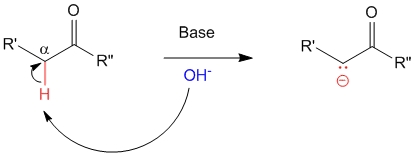
Source Image: pharmaxchange.info
Download Image
SOLVED: Circle-like molecule with the highest equilibrium points) Rank the following molecules by acidity (lowest pKa; highest pKa points) Circle the epibond compound(s) (Meklaeenalann points) Depicted below is a Claisen reaction that 3. Nitro groups are very powerful electron-withdrawing groups. The phenol derivative picric acid (2,4,6 -trinitrophenol) has a pK a of 0.25, lower than that of trifluoroacetic acid. Use a resonance argument to explain why picric acid has such a low pKa. [reveal-answer q=”475315″]Show Solution [/reveal-answer]
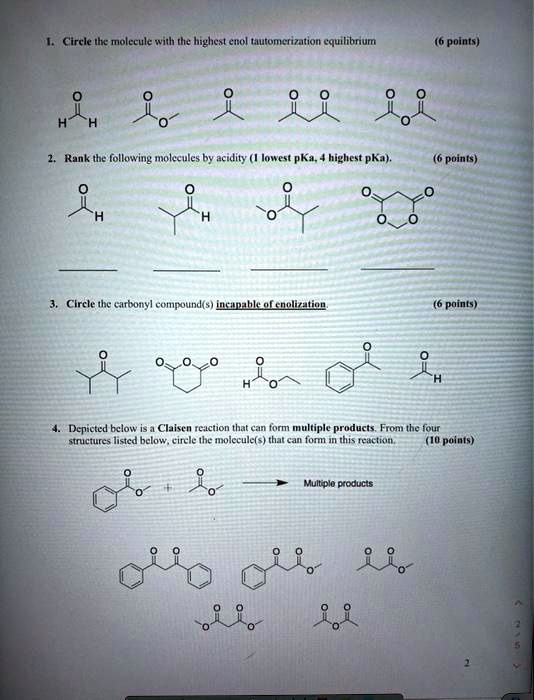
Source Image: numerade.com
Download Image
Organic Chemistry chapter 3 Flashcards | Quizlet Looking at Table 5.2. 1, you see that the pK a of carboxylic acids are in the 4-5 range, the pK a of sulfuric acid is -10, and the pK a of water is 14. Alkenes and alkanes, which are not acidic at all, have pK a values above 30. The lower the pKa value, the stronger the acid. Table 5.2. 1: Representative acid constants.

Source Image: quizlet.com
Download Image
Question on pKa values for amino acids : r/Mcat EXPERT VERIFIED Step 1/9 Step 1: Identify the acids in the given list. The acids in the given list are OH, NH, OH, CI, and 3. Step 2: Determine the pKa values of each acid. The pKa values of the acids are not provided in the given list. Therefore, we cannot directly rank them based on pKa values.

Source Image: reddit.com
Download Image
Which of the following acids has lowest `pK_(a)` value? – YouTube Order the following acids from highest to lowest pKa value. Start with the highest pKa as number. CH3CH3 H2NCH3 HOCH3 HSCH3. 03:42. Without knowing the exact pKa values, rank the compounds A, B, C, and D in order of increasing acidity: Br … Rank the following acids in order of increasing pKa. (1=lowest pKa & 4=highest pKa) OH

Source Image: youtube.com
Download Image
SOLVED: 20.4 points. Rank the following acids from highest pKa to lowest pKa. Explain your reasoning. Sep 11, 2023HO Submitted by Stacy P. Sep. 11, 2023 07:21 a.m. Instant Answer Step 1/3 Step 1: Determine the acidity of each acid based on its structure and functional groups. HO- is a hydroxide ion, which is a strong base and not an acid. Therefore, it does not have a pKa value. Step 2/3
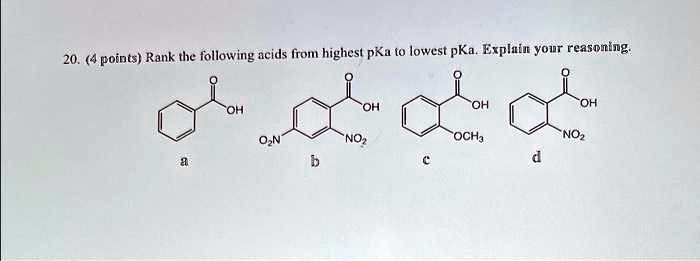
Source Image: numerade.com
Download Image
Which carboxylic acid has highest pKa value? | Filo Predict the Outcome of Organic Acid-Base Reaction — Use pK a as Criterion With the knowledge of acidity and pK a, we are now ready to see how to apply this information to the understanding of organic reactions from an acid-base perspective.. The following reaction is an example in Section 3.2.If you take a closer look at the reactants and products, you will find that the “product” side

Source Image: askfilo.com
Download Image
SOLVED: Circle-like molecule with the highest equilibrium points) Rank the following molecules by acidity (lowest pKa; highest pKa points) Circle the epibond compound(s) (Meklaeenalann points) Depicted below is a Claisen reaction that
Which carboxylic acid has highest pKa value? | Filo Chemistry 10th Edition ISBN: 9781305957404 Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste Publisher: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste Chapter1: Chemical Foundations Section: Chapter Questions Problem 1RQ: Define and explain the differences between the following terms. a. law and theory b. theory and
Question on pKa values for amino acids : r/Mcat SOLVED: 20.4 points. Rank the following acids from highest pKa to lowest pKa. Explain your reasoning. Order the following acids from highest to lowest pKa value. Start with the highest pKa as number. CH3CH3 H2NCH3 HOCH3 HSCH3. 03:42. Without knowing the exact pKa values, rank the compounds A, B, C, and D in order of increasing acidity: Br … Rank the following acids in order of increasing pKa. (1=lowest pKa & 4=highest pKa) OH